Many radiology departments experience an increase in the number of prostate MRIs. Both the EAU (European Association of Urology) and ACR (American College of Radiology) have recently adopted new guidelines that recommend the acquisition of an MRI before obtaining a prostate biopsy. MRI provides valuable information to prevent unnecessary biopsies, and, in case a biopsy cannot be avoided, MR images can support the biopsy procedure by providing the location of suspicious areas. As a result, we are moving towards a world where MRI will play a key role in diagnosing cancer in the prostate. With 1.3M cases yearly, this will lead to a massive increase in the number of prostate MRI exams, all of which require the attention of a radiologist. As prostate MRIs are multiparametric MRIs, they are challenging to read and often require an extensive amount of practice and plenty of time to report on. How can artificial intelligence (AI) provide support?
In this article, we discuss six ways in which AI can assist radiologists while reading prostate MRIs and help them become better and faster at this task with an increasing workload.
1. Automation of prostate volume measurements and PSA density calculation
A patient who is suspected of having prostate cancer, is often given a prostate-specific antigen (PSA) test first. Although this test is not very sensitive,1 it is a fairly easy test to execute providing initial information for the diagnosis. If PSA levels are too high, further examination is required, and according to current guidelines, this means obtaining an MRI. The MRI can then provide the prostate volume, which is needed to calculate PSA density - a more accurate predictor for pathology than a general PSA level.2
The prostate volume can be obtained in various ways. Not uncommon these days is a measurement along the three dimensions, after which a specific formula is applied leading to an estimation of the volume.3 Of course it would be more precise to have prostate segmentation in the T2w sequence of the multiparametric MRI (mpMRI). However, when performed manually, creating such a segmentation can be an extremely time-consuming task. As luck would have it, AI is perfectly suited to fully automatically segment the prostate gland in an MRI image. Hence, a more accurate volume can be calculated, spending even less time than normally necessary with the 3D formula-based estimation method.
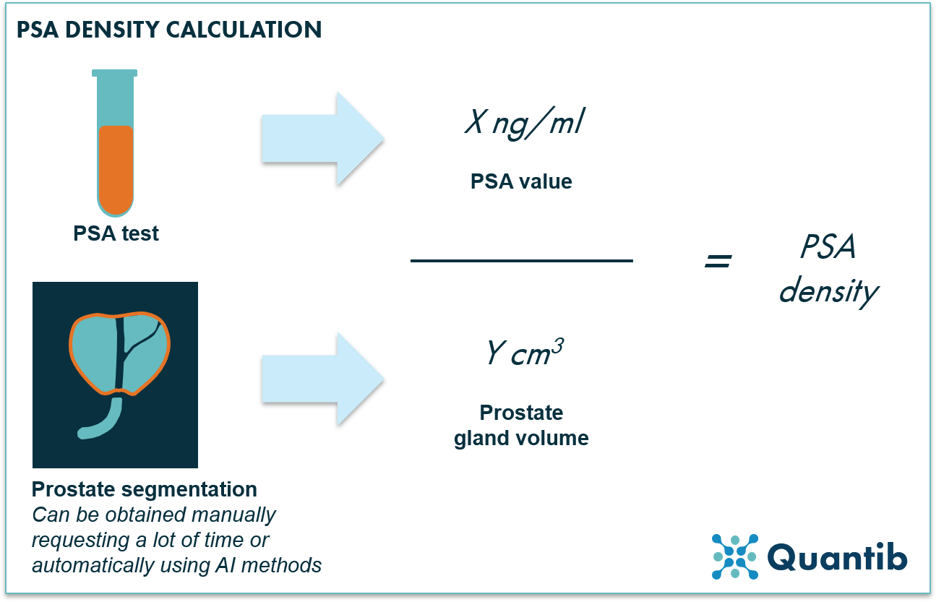
Figure 1: PSA density is calculated by dividing the PSA value by the volume of the prostate gland. The prostate gland volume can be obtained in various ways: manually or automatically, e.g. using AI methods.
An algorithm needs time to run its calculations and obtain a value for prostate volume. If it would take too much time, the added value of the algorithm would diminish. However, very promising results have been shown with processing times of only 10 seconds.4 Additionally, most clinically installed AI solutions start running right after image acquisition; hence, results are in well before a radiologist sits down to analyze the images.
2. Automated lookup of mpMRI prostate prior exams
Although not many AI radiology companies are advertising it, automated lookup of priors can save radiologists a lot of time scrolling through patient lists searching for the right scan obtained at an earlier point in time. Would it not be great if AI would solve this problem with one or two clicks of a button?
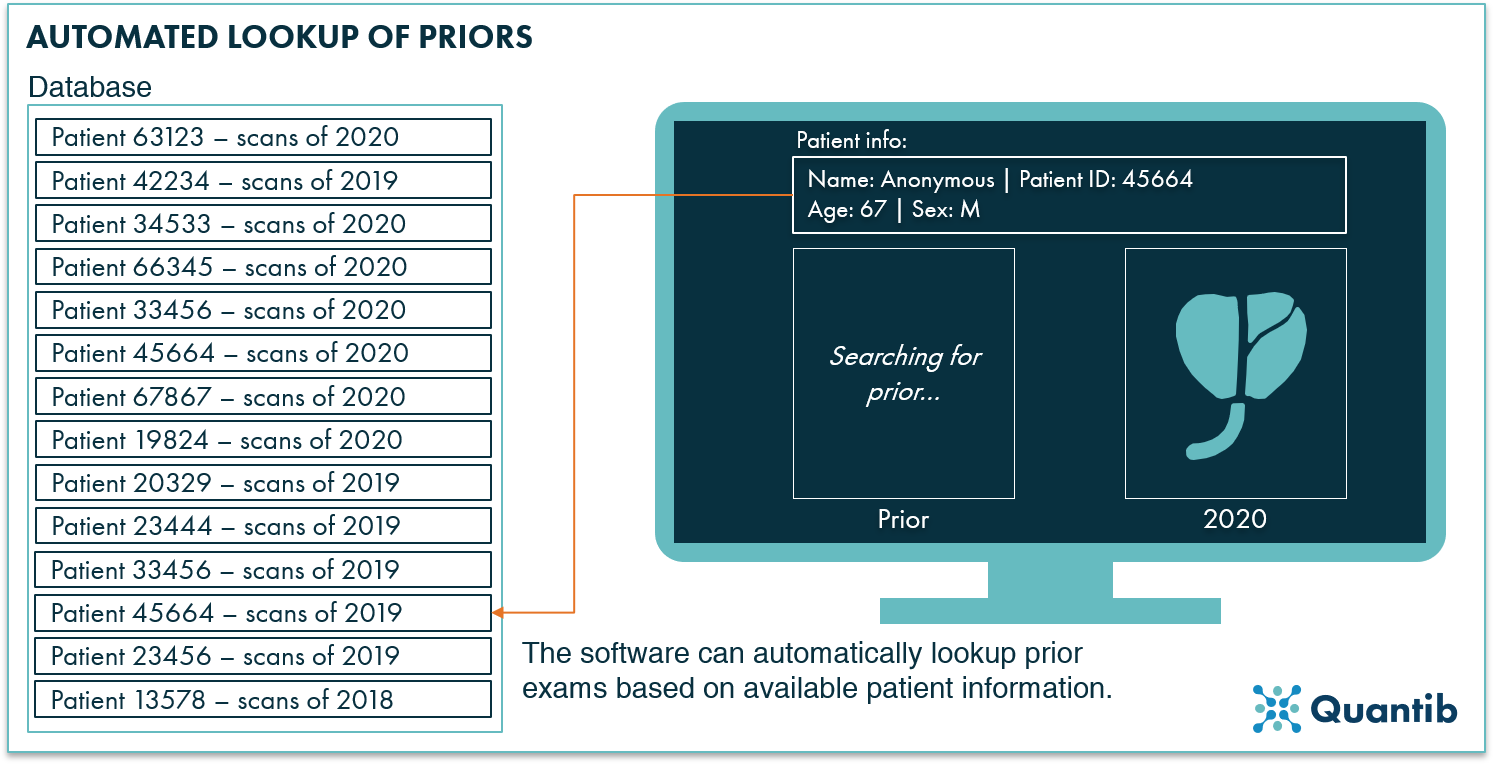
Figure 2: Using patient information (e.g. the patient ID), the AI software can automatically detect priors in the hospital system (e.g. in the PACS).
Looking up priors may seem like a straightforward task. However, since there are many different types of PACS software, RIS programs, etc., from a technical point of view, it is not that easy. If AI radiology companies are eager to include prior lookup functionality in their software, they need to link the algorithms in a fast and efficient way to the systems the hospital already has in place. This is tricky and can be a completely different challenge for each hospital interested in AI analyses.
3. Detection of lesions on mpMRI prostate
The detection of prostate lesions on MRI scans has proven to be a challenging task mostly reserved for the experts in the field,5–7. If AI solutions can guide radiologists to become faster and better at this task, the value added could be substantial.
In practice AI-based detection can mean several things, as is shown in figure 3. The software can pinpoint a specific location of the lesion, or provide a heatmap indicating suspicious areas.8 Another option is fully automated segmentation of all lesions.9 This approach provides volume per lesion, information about the wider location in case the lesion spreads over multiple zones, and insights concerning extracapsular extension. That is, all results that are a valuable input for (AI-assisted) PI-RADS scoring.
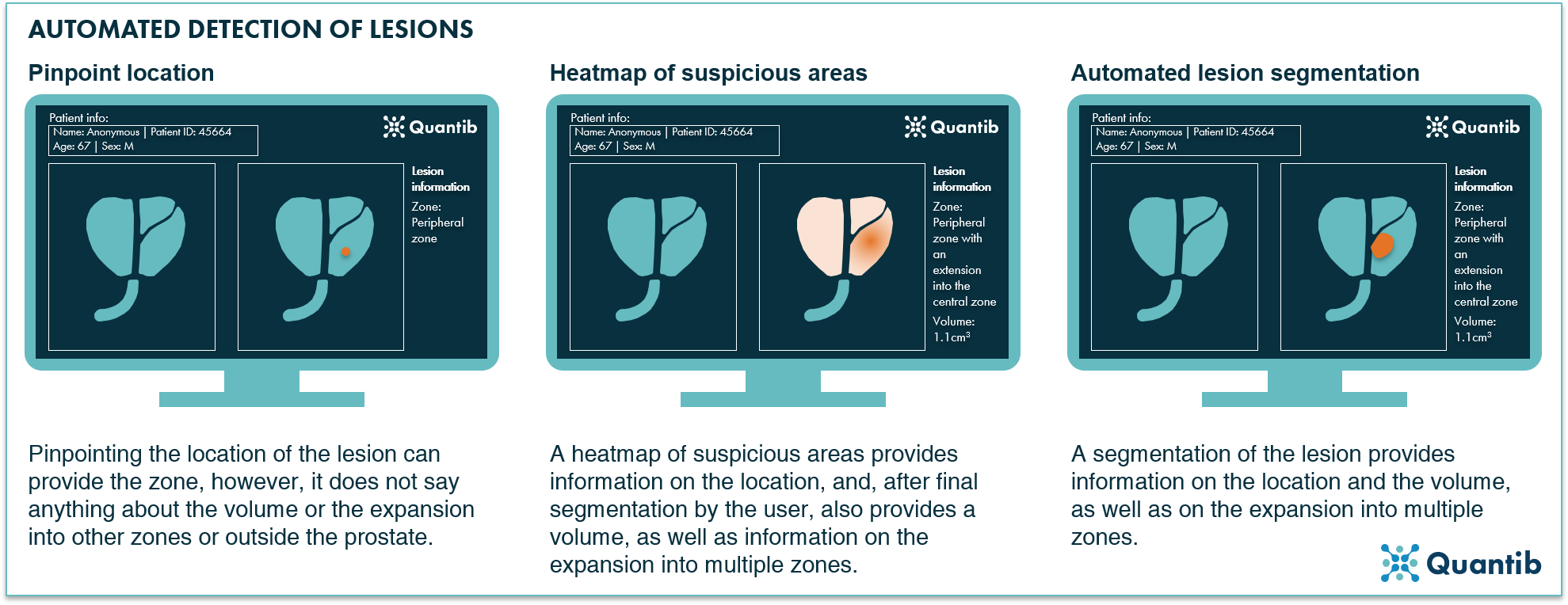
Figure 3: AI radiology software can help detect prostate lesions on mpMRI in several ways. The algorithm can, for example, pinpoint a lesion, provide a heatmap after which the user can determine a final segmentation, or the software can provide a fully automated segmentation.
4. Staging of the lesions on prostate mpMRI
AI support for staging of prostate lesions on MRI scans can be implemented in two ways (see figure 4). Firstly, a separate algorithm can be developed for every sub-question a radiologist answers, such as prostate segmentation into its different zones. Output to all these answers can then be combined to get a final score (such as a PI-RADS score), either by the radiologist or by another algorithm. Alternatively, an overall algorithm can be trained to directly output a score. The second route may sound tempting, but regulatory-wise it is a challenging one. Outputting a PI-RADS score is considered Computer Aided Diagnosis, making the regulatory process more of a hassle than when you provide “just the input” for such a diagnostic verdict.
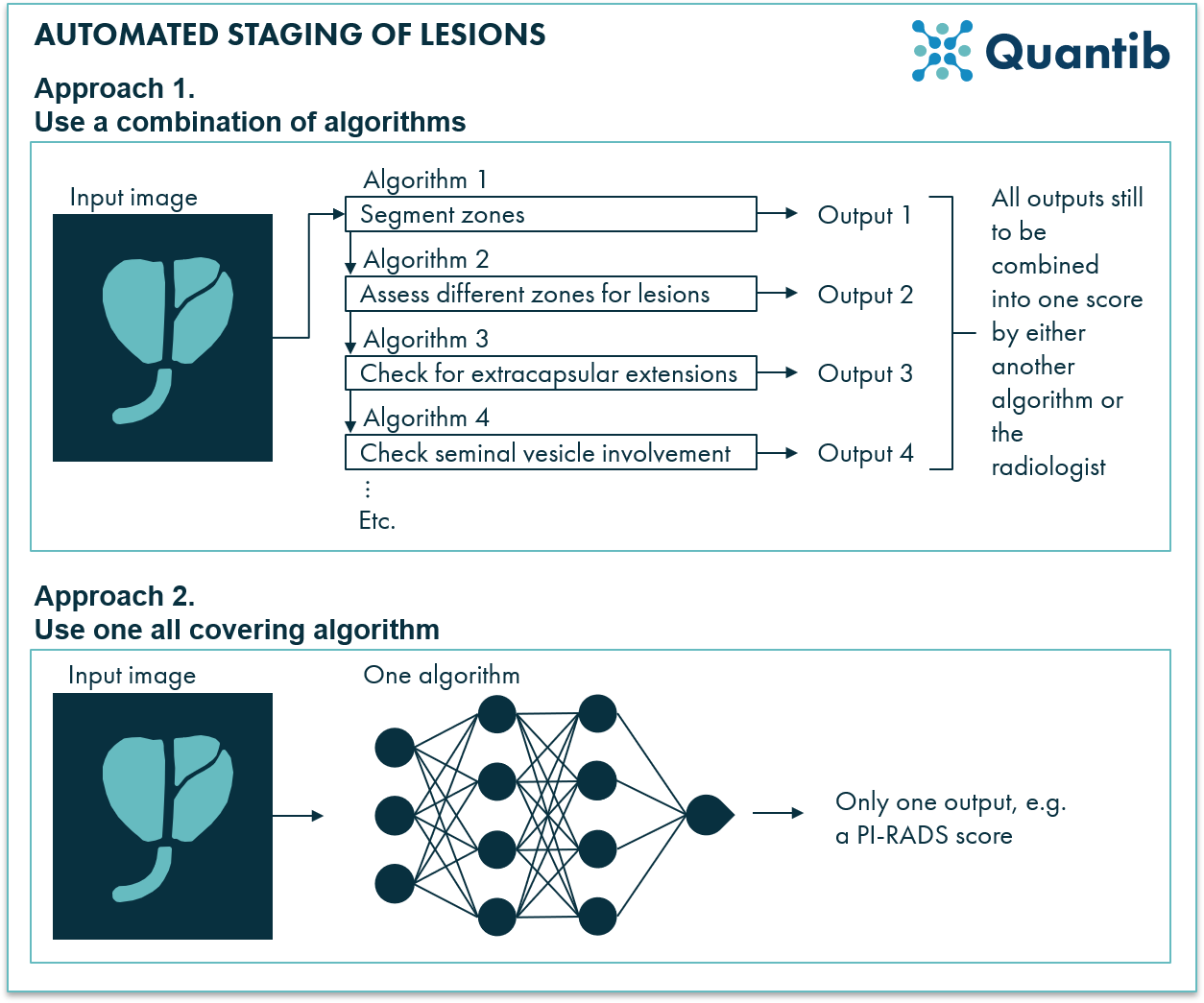
Figure 4: Staging of prostate lesions can be supported in several ways by AI radiology software. An AI program can use multiple algorithms to provide answers to questions relevant for the reading process and then combine these answers into one score, but it is also possible to use only one algorithm to get to a score directly.
For the first approach many different types of algorithms are fit. One can turn to more advanced deep neural networks, but also, depending on the required output, much more simple machine learning approaches may do the trick just fine. For some sub-algorithms, it might be even as simple as intensity thresholding (for example, for finding hypo-intense areas in the peripheral zone, this could be a method that works just fine). The second approach usually requires more advanced techniques. Deep learning methods are usually very suited to predict an outcome such as a PI-RADS score or a lesion significance score. This can be done by using many different features obtained from the input image without going into the hassle of figuring out all these features yourself.
Read about Martina Martins, MD her experience with using AI software in her
prostate MRI reporting.
5. Easy and fast standardized reporting of mpMRI prostate cases
Creating a clear, comprehensive report communicating a precise message to the urologist can take a lot of time. Additionally, style and quality of reports can vary strongly from one radiologist to another. Including a standardized workflow in the AI radiology software will support the reporting process and can help reduce inter- and intra-observer variability. Additionally, the program can offer an intuitive step-by-step roadmap, guiding the user through all mandatory steps such as PSA density calculation, automated zone notification, and a step-wise determination of PI-RADS including questions from the current guidelines, making the process easier and faster. This will all result in an objective report that is clear and easy to comprehend by referring physicians.
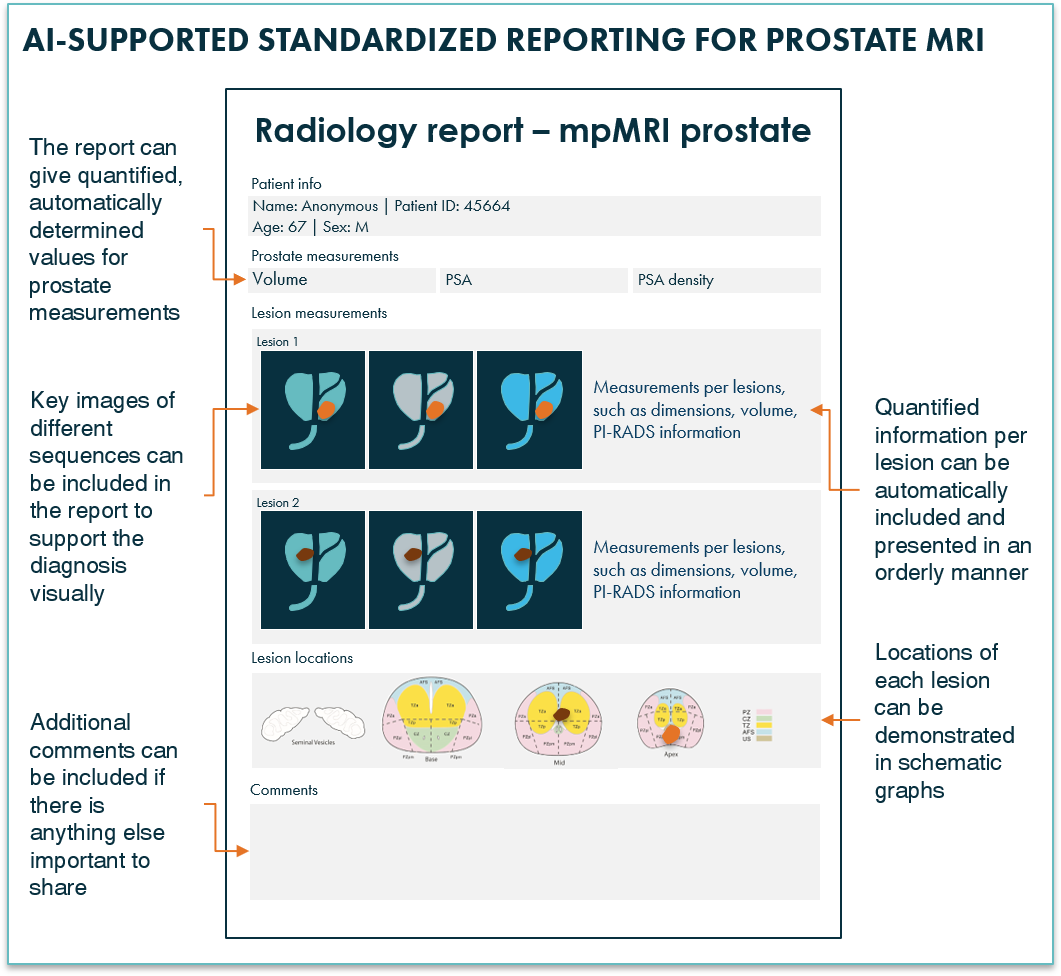
Figure 5: AI radiology software can provide mpMRI prostate reports with clear, complete information.
6. Multidisciplinary team meeting support of mpMRI prostate results
There can be many challenges for radiologists presenting their findings during multidisciplinary team meetings (MDTs), such as having the right images available, pre-selected with the right slice, providing objective and quantified information independent of created the case report , and presenting results in graphs that immediately point out the right information. Using the right AI radiology software to prepare these meetings provides the opportunity to tackle many of these challenges. A more all-round platform for MDT support, for example including pathology results, might even provide the options to integrate non-imaging results, such as Gleason scores.
To conclude
There are many ways in which AI software can support the radiology workflow of reading and reporting prostate MRI scans. When covering a broader scope of the diagnostic pathway, e.g. including pathology findings, possibilities become even more abundant. Curious to learn more about our prostate MRI project? Contact us today!
Bibliography
- Holmström, B., Johansson, M., Bergh, A., Stenman, U. & Hallmans, G. Prostate specific antigen for early detection of prostate cancer: longitudinal study. 339, (2009).
- Sfoungaristos, S. & Perimenis, P. PSA density is superior than PSA and Gleason score for adverse pathologic features prediction in patients with clinically localized prostate cancer. 6, (2012).
- Wasserman, N. F., Niendorf, E. & Spilseth, B. Measurement of Prostate Volume with MRI ( A Guide for the Perplexed): Biproximate Method with Analysis of Precision and Accuracy. Sci. Rep. 55455, 1–12 (2020).
- Fotin, S. V et al. Fully Automated Prostate Segmentation on MRI: Comparison With Manual Segmentation Methods and Specimen Volumes. 720–729 (2015). doi:10.2214/AJR.12.9712
- Steenbergen, P. et al. Prostate tumor delineation using multiparametric magnetic resonance imaging : Inter-observer variability and pathology validation. Radiother. Oncol. 15, (2015).
- Nobin, J. Le, Orczyk, C., Deng, F. & Melamed, J. Prostate tumour volumes: evaluation of the agreement between magnetic resonance imaging and histology using novel co-registration software. 114, 105–112 (2014).
- Ayoola, A. et al. The Learning Curve in Prostate MRI Interpretation: Self-Directed Learning Versus Continual Reader Feedback. 208, 1–9 (2017).
- Lay, N. et al. Detection of prostate cancer in multiparametric MRI using random forest with instance weighting. 4, (2017).
- Liu, X. et al. Prostate Cancer Segmentation With Simultaneous Estimation of Markov Random Field Parameters and Class. 28, 906–915 (2009).
