Prostate Cancer (PCa) has the second highest incidence in men worldwide1, affecting more than 1.4 million men, and has the highest expected incidence in men in the US2 , with more than 288.000 expected new cases this year.
Due to the challenge that PCa presents because of its growing incidence, early detection is crucial as it has been established that an early diagnosis can lead to a 97% survival rate of the patients3. In order to make early diagnosis a reality, it is imperative to find ways of making the PCa clinical pathway more efficient, whether that is by introducing screening programs or by optimizing the diagnostic and treatment processes themselves.
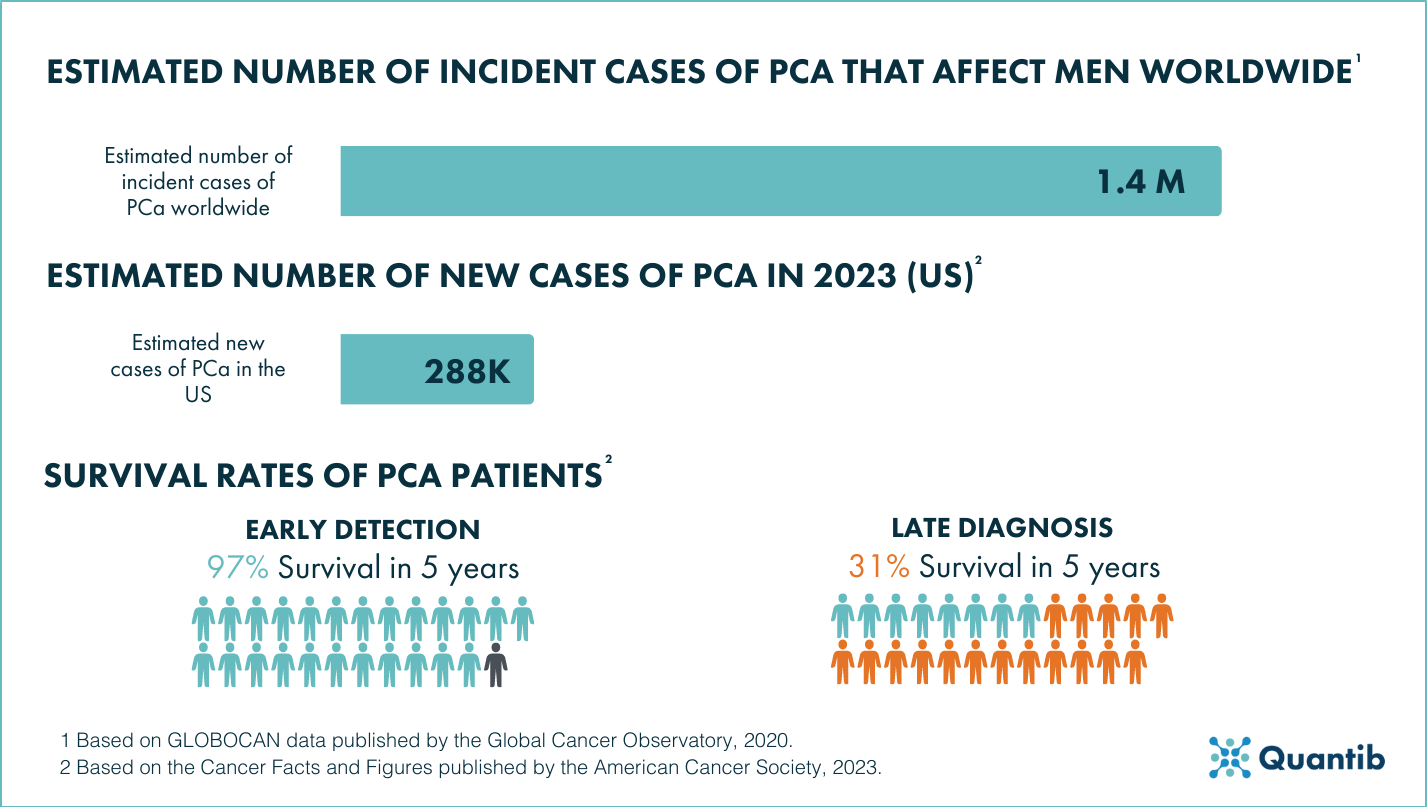
Figure 1: The challenge of Prostate Cancer 1 2 3.
And that is where artificial intelligence (AI) comes in. AI is one of the technologies that has been thoroughly researched in order to achieve innovations in the field of prostate oncology and healthcare in general.
In this article we have compiled some of the ways that AI can be applied to the PCa clinical pathway, some of them can be implemented in clinical practice as they are commercially available and some are still being nurtured in the academic field as they have yet to reach maturity.
AI applications to the diagnosis of Prostate Cancer
Research has shown that AI can actually be applied to multiple procedures that are part of the diagnostic workup of PCa.
AI and PSA testing
Even though AI cannot be applied to PSA testing itself, the information gathered by the PSA test can be used in combination with AI and other patient information to predict the risk of the patient getting a positive prostate biopsy4. In a recent study, the use of machine learning (ML) was proven to improve risk stratification of PCa compared to the use of just biochemical parameters such as PSA5.
AI and Imaging
Even though there are multiple options available when it comes to prostate imaging, the applications of AI in imaging are quite similar. These AI applications range from prostate gland, sub-region or lesion segmentation to (semi)automated lesion detection or scoring of the lesion6. There are also AI algorithms aimed at evaluating the quality of the scans7 and even improving that quality, for example by reducing artifacts and noise8.
AI and prostate MRI
The diagnostic read of magnetic resonance imaging (MRI) of the prostate is complex and involves many steps, so it offers a wide range of opportunities for the application of AI. It has been proven that AI can be applied to the automation of prostate volume measurements9–11 and PSA density calculation. Besides that, there has been ample research that proved the successful application of AI to prostate gland and region segmentation12, as well as lesion segmentation13, lesion detection 14,15 and grading of lesions according to PI-RADS16.
MRI is one of the imaging modalities in which these AI applications have reached the market the most as there are multiple AI software currently available that cover one or more of these applications17. When developed into fully functional software for clinical use, they can take two main shapes: automated, in which the software will generate output without interaction of the radiologist or semi-automated, in which the software allows radiologists to adjust any of the results and make the last call on their MRI read.
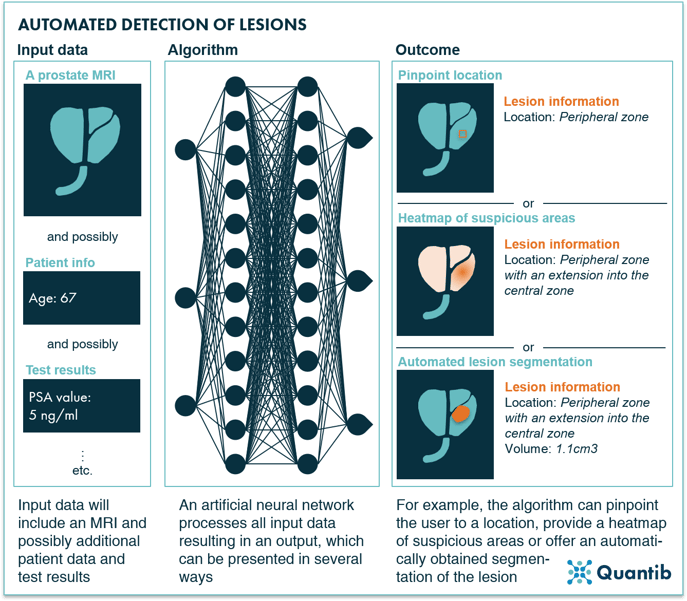 Figure 2: AI-driven automated detection of lesions based on prostate MRI.
Figure 2: AI-driven automated detection of lesions based on prostate MRI.
There are clinical validation studies published for commercially available algorithms aimed at evaluating if such algorithms do indeed provide value to the diagnostic pathway of PCa by investigating the effects that it has on the accuracy, variability and reporting times of the user18–21.
Do you want to know more about AI software applied to prostate MRI reading? Discover Quantib® Prostate.
AI and other imaging modalities of the prostate
When it comes to ultrasound (US) and position emission tomography computed tomography (PET/CT) images of the prostate, some of the successful AI applications that have been researched are automated prostate gland22–26 and region segmentation27, ROI detection28,29 and lesion detection in dynamic images30.
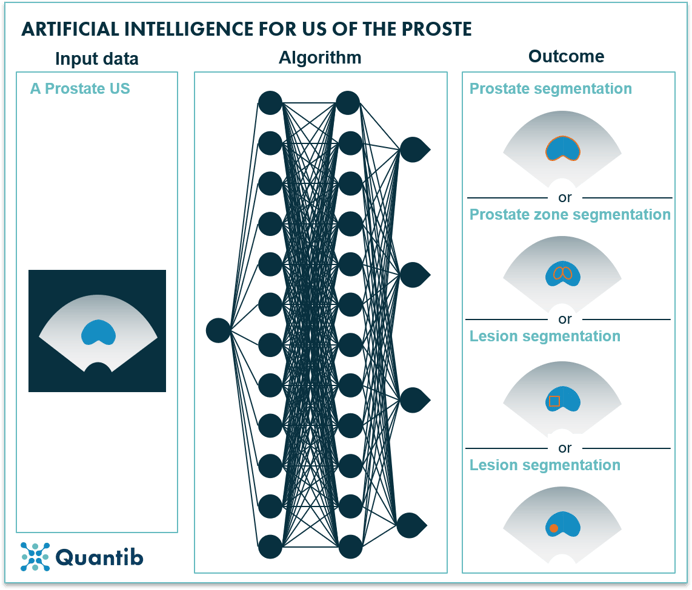 Figure 3: AI for prostate ultrasound.
Figure 3: AI for prostate ultrasound.
A newer imaging modality has been vastly researched as of lately to assess its value to the prostate cancer clinical pathway: the prostate-specific membrane antigen positron emission tomography (PSMA/PET)31. The research of AI applications is a little bit more limited in this imaging modality but, for example, researchers have created an AI method for PCa tumor and metastases detection32.
The continuous research regarding the efficacy and potential uses of the different imaging modalities previously mentioned make it difficult to establish which imaging modality can be considered “best” for the PCa clinical pathway. However, due to said research, more international organizations, such as the European Association of Urology (EAU) and the American College of Radiology (ACR) have adapted their guidelines to set the acquisition of a prostate MRI pre-biopsy as the recommended practice33,34.
Besides the benefits that these AI applications can have to the diagnostic read of these imaging modalities, like a higher accuracy, decreased inter-observer variability and reduced reporting times, the output provided by the radiologist, primarily the segmentations and standardized reports, can have a positive impact in the rest of the diagnostic and clinical pathway of PCa. The results can be used for image-guided diagnostic processes and treatments, like MRI guided biopsies or radiotherapy, or for better risk stratification and treatment planning6.
AI applications to Prostate Cancer pathology
The application of AI to histopathological images in the field of PCa has also been vastly researched, specifically when it comes to tumor detection and tumor grading. AI algorithms for tumor detection have reached a high level of accuracy when discerning between benign and malignant tissue derived from prostate biopsies35,36. When it comes to tumor grading, research shows that AI algorithms can increase inter-observer agreement and it decreased the amount of missed tumors compared to pathologists37,38.
AI applications to the treatment Prostate Cancer
Lastly, researchers have also evaluated the application of AI to the treatment of PCa, from the selection of a specific treatment based on the input gathered during the diagnosis process39,40 to dosage calculation for radiotherapy or brachytherapy treatments41,42 and surgical support through the use of augmented reality or surgical robots43.
AI for the active surveillance of Prostate Cancer
In some cases of PCa, active surveillance can be the best option to proceed. In these cases, it is normal for patients to have regular imaging procedures to track the progression of the lesions and reevaluate if treatment is necessary. The already mentioned applications of AI to prostate imaging can be very helpful for this as some algorithms might allow the comparison of quantitative values from scans taken at different time points. On the other hand, there is also research surrounding the use of AI models to predict the progression of the tumor grade while the patients are on active surveillance44.
Conclusion
There is the wide variety of options to apply AI in the prostate cancer pathway. Some of them have found a way into the clinical setting and some other are still something to look out for in the future, but through all the academic research and clinical validation, there is no doubt that AI can add substantial value to the entire clinical pathway of PCa.
Read an in-depth guide about AI and its application to Prostate Cancer oncology in our Ultimate Guide
Bibliography
- Global Cancer Observatory. Cancer today: Estimated number incident cases and deaths worldwide, males, all ages. International Agency for Research on Cancer https://gco.iarc.fr/today (2021).
- Siegel, R. L., Miller, K. D., Wagle, N. S. & Jemal, A. Cancer statistics, 2023. CA Cancer J Clin 73, 17–48 (2023).
- American Cancer Society. Cancer facts and figures 2023. (2023).
- Stephan, C. et al. Multicenter Evaluation of an Artificial Neural Network to Increase the Prostate Cancer Detection Rate and Reduce Unnecessary Biopsies. Clin Chem 48, 1279–1287 (2002).
- Perera, M. et al. PSA-based machine learning model improves prostate cancer risk stratification in a screening population. World J Urol 39, 1897–1902 (2021).
- Six, O., Veldhuis, W. & Akin, O. The ultimate guide to AI in prostate cancer. https://www.quantib.com/the-ultimate-guide-to-ai-in-prostate-cancer (2021).
- Giganti, F. et al. Multiparametric prostate MRI quality assessment using a semi-automated PI-QUAL software program. Eur Radiol Exp 5, 48 (2021).
- Wang, X. et al. Novel deep learning-based noise reduction technique for prostate magnetic resonance imaging. Abdominal Radiology 46, 3378–3386 (2021).
- Bardis, M. D. et al. Applications of Artificial Intelligence to Prostate Multiparametric MRI (mpMRI): Current and Emerging Trends. Cancers (Basel) 12, 1204 (2020).
- Lee, D. K. et al. Three-Dimensional Convolutional Neural Network for Prostate MRI Segmentation and Comparison of Prostate Volume Measurements by Use of Artificial Neural Network and Ellipsoid Formula. American Journal of Roentgenology 214, 1229–1238 (2020).
- Thimansson, E. et al. Deep learning algorithm performs similarly to radiologists in the assessment of prostate volume on MRI. Eur Radiol 33, 2519–2528 (2022).
- Belue, M. J. & Turkbey, B. Tasks for artificial intelligence in prostate MRI. Eur Radiol Exp 6, 33 (2022).
- Gunashekar, D. D. et al. Explainable AI for CNN-based prostate tumor segmentation in multi-parametric MRI correlated to whole mount histopathology. Radiation Oncology 17, 65 (2022).
- Bonekamp, D. et al. Radiomic Machine Learning for Characterization of Prostate Lesions with MRI: Comparison to ADC Values. Radiology 289, 128–137 (2018).
- Lay, N. et al. Detection of prostate cancer in multiparametric MRI using random forest with instance weighting. Journal of Medical Imaging 4, 024506 (2017).
- Sanford, T. et al. Deep‐Learning‐Based Artificial Intelligence for <scp>PI‐RADS</scp> Classification to Assist Multiparametric Prostate <scp>MRI</scp> Interpretation: A Development Study. Journal of Magnetic Resonance Imaging 52, 1499–1507 (2020).
- van Leeuwen, K. Abdomen MR products. AI for radiology https://grand-challenge.org/aiforradiology/?subspeciality=Abdomen&modality=MR&ce_under=All&ce_class=All&fda_class=All&sort_by=last%20modified&search= (2023).
- Faiella, E. et al. Quantib Prostate Compared to an Expert Radiologist for the Diagnosis of Prostate Cancer on mpMRI: A Single-Center Preliminary Study. Tomography 8, 2010–2019 (2022).
- Cipollari, S. et al. Biparametric prostate MRI: impact of a deep learning-based software and of quantitative ADC values on the inter-reader agreement of experienced and inexperienced readers. Radiol Med 127, 1245–1253 (2022).
- Forookhi, A. et al. Bridging the experience gap in prostate multiparametric magnetic resonance imaging using artificial intelligence: A prospective multi-reader comparison study on inter-reader agreement in PI-RADS v2.1, image quality and reporting time between novice and expert readers. Eur J Radiol 161, 110749 (2023).
- Turkbey, B. & Haider, M. A. Artificial Intelligence for Automated Cancer Detection on Prostate MRI: Opportunities and Ongoing Challenges, From the AJR Special Series on AI Applications. American Journal of Roentgenology 219, 188–194 (2022).
- Harvey, C. J., Pilcher, J., Richenberg, J., Patel, U. & Frauscher, F. Applications of transrectal ultrasound in prostate cancer. Br J Radiol 85, S3–S17 (2012).
- Kachouie, N. N. & Fieguth, P. A Medical Texture Local Binary Pattern For TRUS Prostate Segmentation. in 2007 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society 5605–5608 (IEEE, 2007). doi:10.1109/IEMBS.2007.4353617.
- Tutar, I. B. et al. Semiautomatic 3-D Prostate Segmentation from TRUS Images Using Spherical Harmonics. IEEE Trans Med Imaging 25, 1645–1654 (2006).
- Badiei, S., Salcudean, S. E., Varah, J. & Morris, W. J. Prostate Segmentation in 2D Ultrasound Images Using Image Warping and Ellipse Fitting. in 17–24 (2006). doi:10.1007/11866763_3.
- Mortensen, M. A. et al. Artificial intelligence‐based versus manual assessment of prostate cancer in the prostate gland: a method comparison study. Clin Physiol Funct Imaging 39, 399–406 (2019).
- van Sloun, R. J. G. et al. Deep Learning for Real-time, Automatic, and Scanner-adapted Prostate (Zone) Segmentation of Transrectal Ultrasound, for Example, Magnetic Resonance Imaging–transrectal Ultrasound Fusion Prostate Biopsy. Eur Urol Focus 7, 78–85 (2021).
- Mohamed, S. S., Li, J., Salama, M. M. A. & Freeman, G. Prostate Tissue Texture Feature Extraction for Suspicious Regions Identification on TRUS Images. J Digit Imaging 22, 503–518 (2009).
- Wildeboer, R. R. et al. Automated multiparametric localization of prostate cancer based on B-mode, shear-wave elastography, and contrast-enhanced ultrasound radiomics. Eur Radiol 30, 806–815 (2020).
- Rubinstein, E. et al. Unsupervised tumor detection in Dynamic PET/CT imaging of the prostate. Med Image Anal 55, 27–40 (2019).
- Tsechelidis, I. & Vrachimis, A. PSMA PET in Imaging Prostate Cancer. Front Oncol 12, (2022).
- Trägårdh, E. et al. Freely Available, Fully Automated AI-Based Analysis of Primary Tumour and Metastases of Prostate Cancer in Whole-Body [18F]-PSMA-1007 PET-CT. Diagnostics 12, 2101 (2022).
- Mottet, N. et al. EAU Guidelines. (2020).
- ACR. Prostate MRI model policy. (2019).
- Ayyad, S. M. et al. Role of ai and histopathological images in detecting prostate cancer: A survey. Sensors vol. 21 Preprint at https://doi.org/10.3390/s21082586 (2021).
- Van Booven, D. J. et al. A systematic review of artificial intelligence in prostate cancer. Research and Reports in Urology vol. 13 31–39 Preprint at https://doi.org/10.2147/RRU.S268596 (2021).
- Bulten, W. et al. Artificial intelligence assistance significantly improves Gleason grading of prostate biopsies by pathologists. Modern Pathology 34, (2021).
- Bulten, W. et al. Artificial intelligence for diagnosis and Gleason grading of prostate cancer: the PANDA challenge. Nat Med (2022) doi:10.1038/s41591-021-01620-2.
- Yu, S. H. et al. Early experience with Watson for Oncology: a clinical decision-support system for prostate cancer treatment recommendations. World J Urol 39, 407–413 (2021).
- Rabaan, A. A. et al. Artificial Intelligence for Clinical Diagnosis and Treatment of Prostate Cancer. Cancers (Basel) 14, 5595 (2022).
- Ma, M., Kovalchuk, N., Buyyounouski, M. K., Xing, L. & Yang, Y. Dosimetric features‐driven machine learning model for DVH prediction in VMAT treatment planning. Med Phys 46, 857–867 (2019).
- Nicolae, A. et al. Evaluation of a Machine-Learning Algorithm for Treatment Planning in Prostate Low-Dose-Rate Brachytherapy. International Journal of Radiation Oncology*Biology*Physics 97, 822–829 (2017).
- Goldenberg, S. L., Nir, G. & Salcudean, S. E. A new era: artificial intelligence and machine learning in prostate cancer. Nat Rev Urol 16, 391–403 (2019).
- Nayan, M. et al. A machine learning approach to predict progression on active surveillance for prostate cancer. Urologic Oncology: Seminars and Original Investigations 40, 161.e1-161.e7 (2022).