Radiology, the use of imaging modalities and processes to image the human body for diagnostic and treatment purposes has historically been at the forefront of applying the latest technological advancements. Over the previous decade, we have seen an incredible evolution in CT and MR imaging technology, complemented by a recent transformation through artificial intelligence (AI)-based software, expanding the horizon of image quality and diagnostic efficiency.
With the end of 2021 slowly approaching, we look back on this year and the innovations in radiology that will undoubtedly redraw the clinical landscape for the coming years, such as photon-counting CT or point-of-care ultrasonography.
1. Breaching the technical limits of CT imaging
2021 saw European and FDA clinical clearance for the world’s first photon-counting CT (PCCT) scanner1. This technology enables radical improvements in resolution while reducing radiation dose, breaching the previous technical limits of conventional CT. While this technology has been in development for over 15 years, a recent review2 highlights the potential of this technology to dramatically improve clinical CTs.
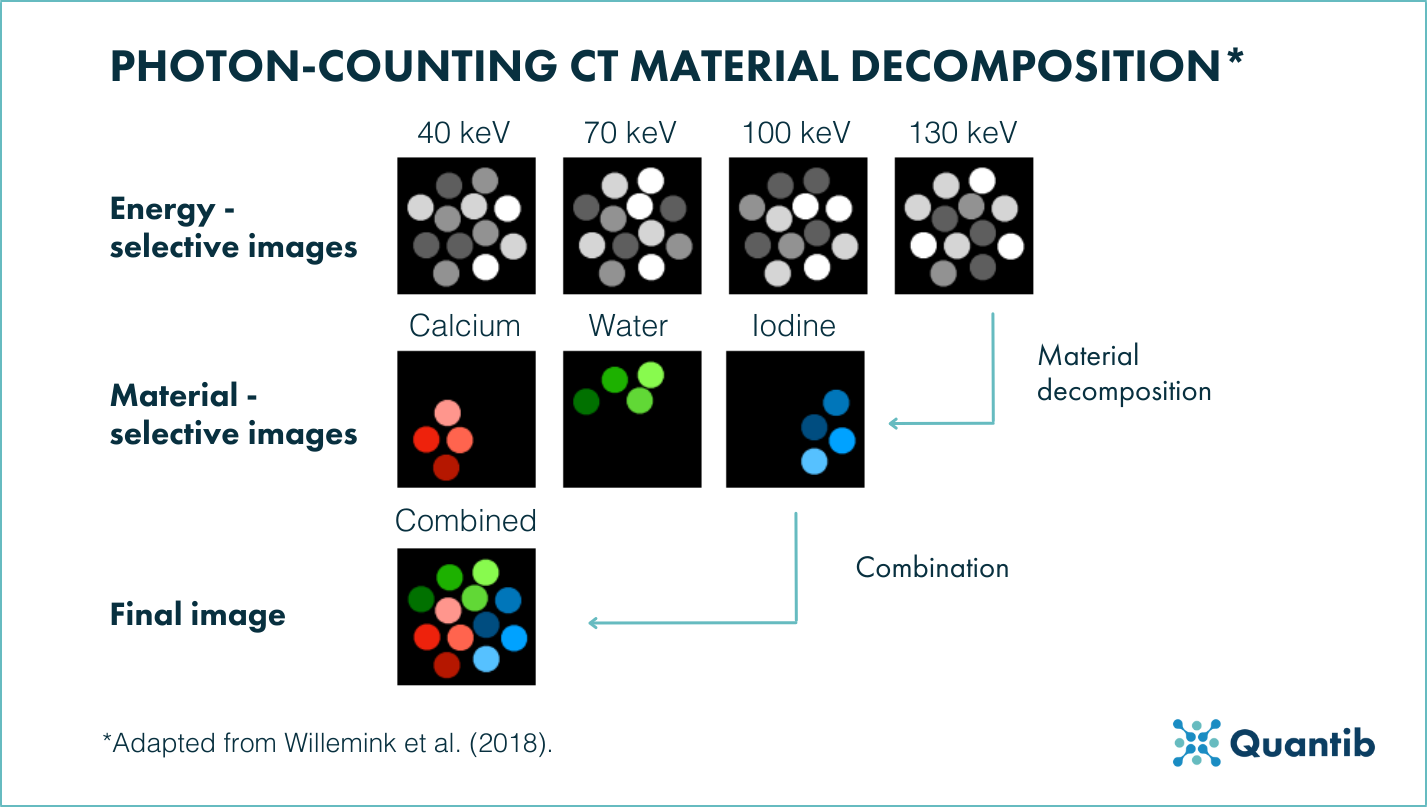
Figure 1. Photon-counting CT material decomposition. Energy-selective images are generated from number of counts in each energy bin. From these images, a data-processing method called material decomposition generates material-selective images. Combining these images generates the final image shown to radiologists2.
Current CT technology is constrained by several limitations, including a trade-off between ionizing radiation dose and image quality. With reductions in dose, artefacts such as image noise and distortions increase, worsening the already poor ability for CT to accurately differentiate between healthy and pathologic tissues. While contrast agents and dual-energy CT scanners aim to counter these issues of tissue differentiation and ambiguous x-ray attenuation values, current energy-integrating detectors (EIDs) can only measure the total deposited energy of x-rays and thus limit spatial resolution. In contrast, photon-counting detectors (PCDs) can convert individual photons directly into an electrical signal and therefore significantly increase spatial resolution and reduce image artefacts. The addition of K-edge imaging (step-change attenuation of x-ray energy for high atomic number elements) also greatly improves the precision of contrast agent concentration and may enable simultaneous quantification of multiple contrast agents.
Although thought to need 5-10 years for its clinical introduction, the first PCCT has entered the market only 3 years after the review was initially published. Its current (clinical) applications mirror those predicted by this research2, specifically breast3, temporal bone4, lung5, and cardiovascular CTs6.
The wide clinical applications and benefits of photon-counting CT also open up a wealth of opportunities to expand the current armamentarium of contrast agents, enabling the use of K-edge metals such as gold nanoparticles and gadolinium which target specific (early) cancer-related biological processes and thus improve diagnosis7. This opportunity can further be enhanced by multiphase scanning with multiple contrast agents all within a single scan.
In regards to anticipated future developments of photon-counting CT, research7 highlights the complementary role artificial intelligence (AI) and deep learning may take on for noise reduction during multiphase scans or scans with a reduced contrast load. Nevertheless, PCCT is only in its infancy, having just received clinical approval, and how this technology will translate to improved clinical outcomes will transpire in the coming years.
2. Growing impact of artificial intelligence
The exponential impact of artificial intelligence (AI) in the field of (neuro)radiology is ever-present. With a higher level of maturity and adoption of this technology, AI is enabling radical shifts in workflow optimization and clinical decision support; enabling radiologists to make more efficient and accurate diagnoses.
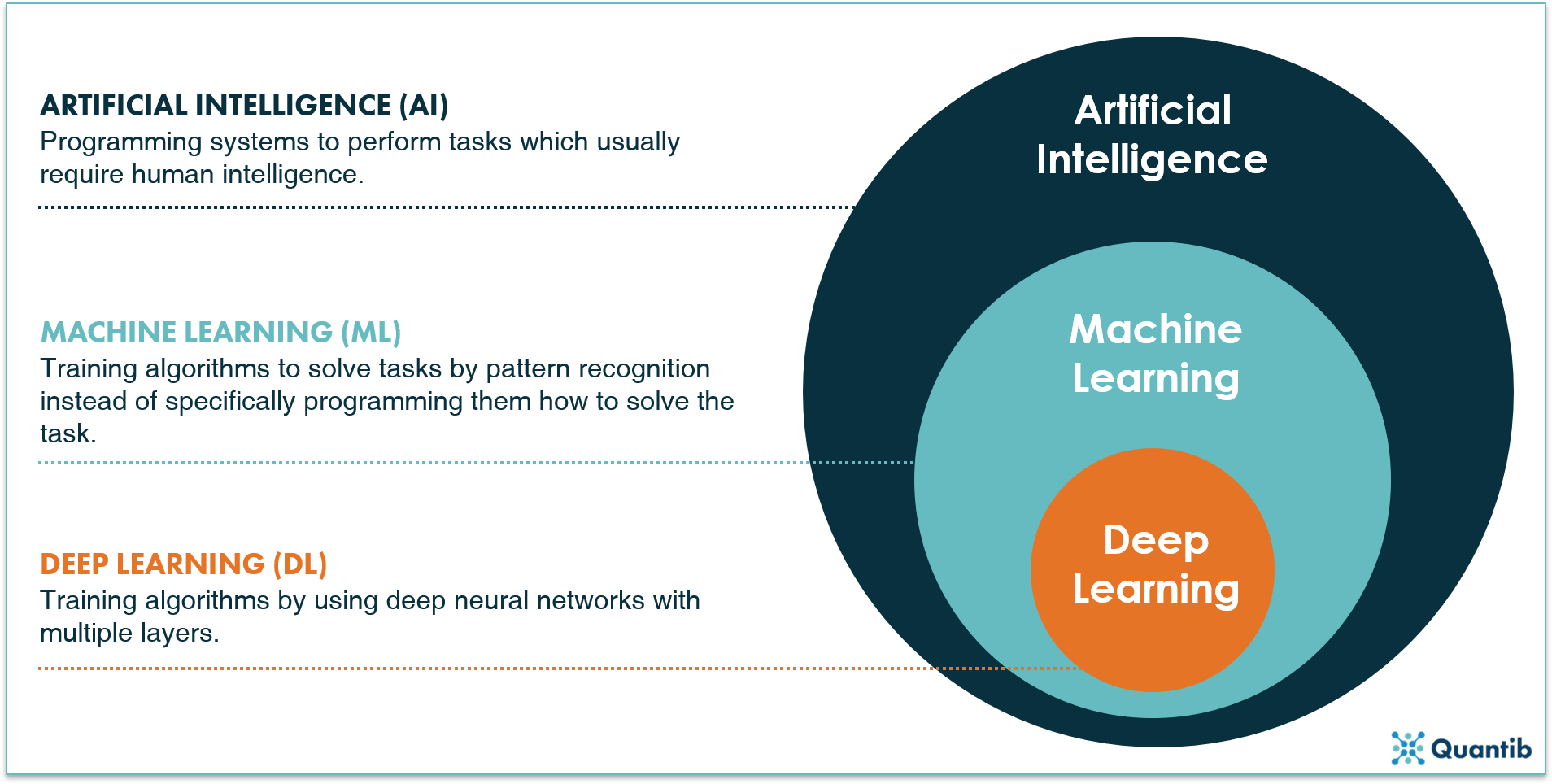
Figure 2. Classification of artificial intelligence architectures8.
For a clear overview of the latest developments and applications, a recent systematic review8 is a useful summary of where the technology currently stands. The authors highlight the adaptable utilization of AI, and specifically deep learning (DL) for a range of imaging modalities - MRI, functional MRI, CT, PET, and US - and neurological disease - Alzheimer disease, vascular injuries, fetal brain development, and cerebral neoplasms.
A primary focus of AI utility was placed on MRI applications, using DL algorithms for image classification (i.e. of major intracranial tumor types9), segmentation (I.e. of anatomical structures of the inner ear10), generation (i.e, of MPRAGE images from mGRE images11), detection (i.e. of neuroanatomical changes in individuals with autism spectrum disorder12), reconstruction (i.e. of high-resolution MRI from under-sampled MRI13), or prediction (i.e. of brain age14). This plethora of applications also extend to the other imaging modalities and demonstrates the incredible heterogeneity of AI applications in (neuro)radiology.
The authors do note that there remains a level of disconnect between AI scientists and physicians, caused by the emphasis on research-based, proof-of-concept DL models rather than providing clinical validation. For an effective integration in clinical practice, the authors advocate for greater collaboration between researchers, physicians, and computer scientists, thus enabling DL to play a major role in imaging, diagnosis, prognosis, treatment, and follow-up of neurologic diseases.
Quantib® ND: an AI software for clinical application in neuroradiology
Quantib® ND, leveraging the power of AI, provides accurate single-time-point and longitudinal quantification of brain atrophy and white matter hyperintensities (WMHs) both related to diseases like dementia and MS. The results and segmentations provided by the software are automatically sent to the PACS of the clinical practice ensuring a seamless integration into the neuroradiologist workflow.
3. Bed-side imaging for improved diagnoses
The COVID-19 pandemic continues to demand efficient and accurate imaging modalities for diagnosis and progression monitoring of critically ill virus patients. For much of the pandemic, CT was heralded as the front line imaging modality of choice, yet the recent growth of point-of-care ultrasonography (PoCUS) ushers in the advent of smaller and more affordable ultrasound machines, capable of accurate and time-efficient diagnosis and monitoring. While PoCUS already garnered sizeable interest prior to COVID-19, a recent review15 highlights the key trends of the technology, its advances in clinical applications, and its increased utility in primary imaging specialties.
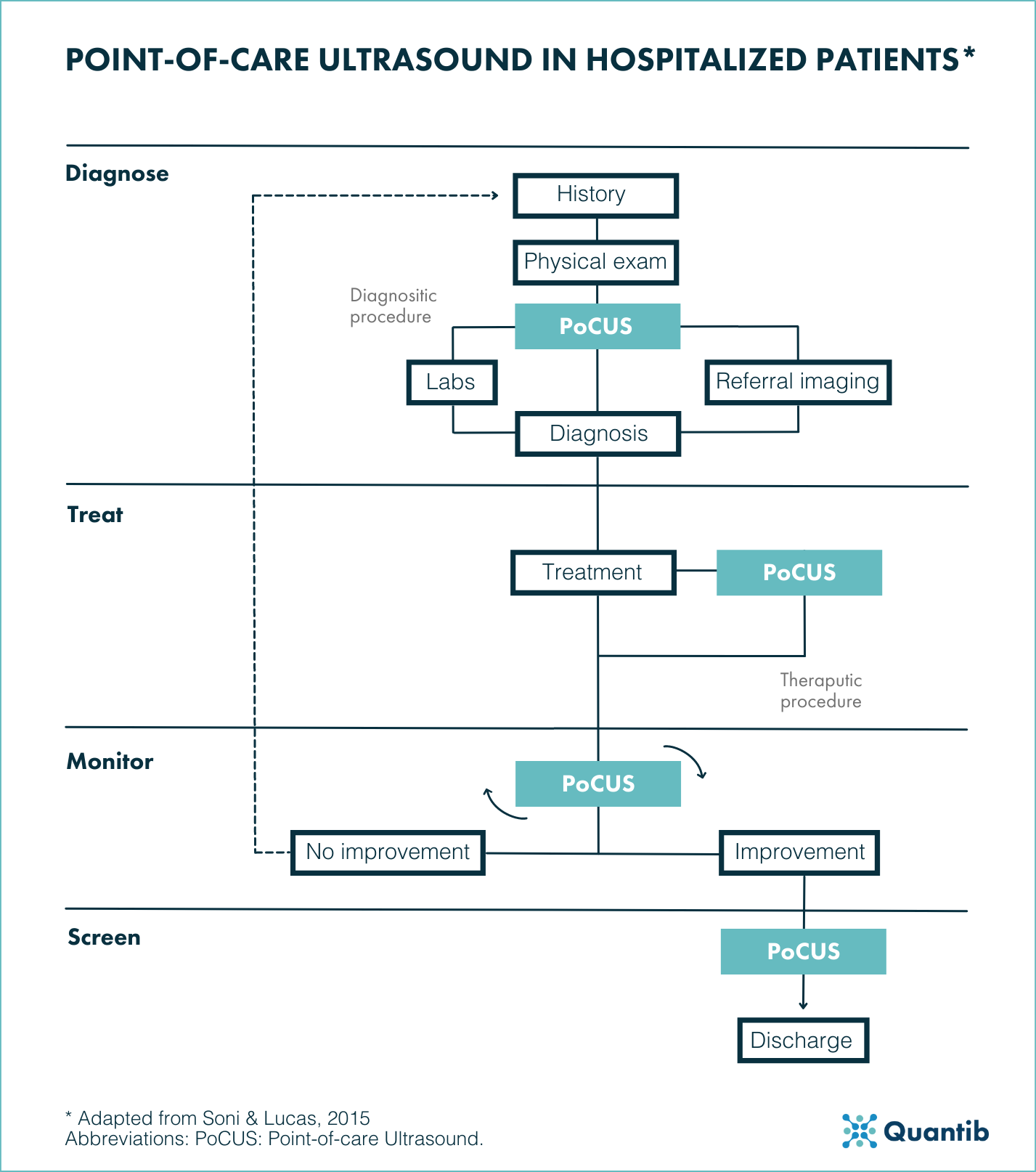
Figure 3. Point-of-care ultrasound in hospitalized patients. Diagnosis: Following initial anamnesis and a physical examination PoCUS may assist in facilitating a diagnosis or guiding further diagnostic workup. PoCUS as part of the therapeutic procedure aim to reduce complications. Furthermore, serial PoCUS exams are performed to monitor disease processes and treatment effects. Lastly, PoCUS is often used in screening for asymptomatic and potentially treatable conditions16. Abbreviations: PoCUS: Point-of-care Ultrasound.
PoCUS enables “acquisition, interpretation, and immediate clinical integration of ultrasonographic imaging performed by a treating clinician at the patient’s bedside rather than by a radiologist or cardiologist"15. To this end, these low-cost hand-held systems are proving readily available to the front-line clinician, assisting in confirming suspected clinical diagnoses or supporting changes in diagnosis. To further optimize this, AI and deep-learning approaches are increasingly implemented in clinical decision-making processes, providing information on, for example, left and right ventricular function, prediction of severity of acute lung disease, or prescriptive imaging guidance, and many others. All these images are ultimately stored in a generic format (ie. DICOM) facilitating standard documentation for quality assurance and billing processes.
Nevertheless, certain questions remain unanswered about PoCUS, including what specific training is necessary to gain sufficient technical competence for clinicians as well as the extent to which the technique results in improved patient functional status, morbidity, and mortality. While research is underway to answer these questions, preliminary results from the substantially increased recent utilization, highlight the potential of this tool of becoming a standard for the frontline clinician.
Conclusion
2021 once again proves that (neuro)radiology remains at the forefront of technological advancements in the healthcare sector. From the clinical implementation of radical new technologies, such as photon-counting CT machines, or the promising developments of tools such as PoCUS, both of which are complemented by the incredible potential of artificial intelligence, current research is ushering in the advent of new efficient and cost-effective technologies.
Bibliography
1. Siemens Healthineers. Siemens Healthineers launches world’s first CT scanner with photon-counting technology. . https://www.siemens-healthineers.com/press/releases/naeotomalpha (2021).
2. Willemink, M. J., Persson, M., Pourmorteza, A., Pelc, N. J. & Fleischmann, D. Photon-counting CT: Technical Principles and Clinical Prospects. Radiology 289, 293–312 (2018).
3. Berger, N. et al. Dedicated Breast Computed Tomography With a Photon-Counting Detector. Investigative Radiology 54, 409–418 (2019).
4. Rajendran, K. et al. Dose Reduction for Sinus and Temporal Bone Imaging Using Photon-Counting Detector CT With an Additional Tin Filter. Investigative Radiology 55, 91–100 (2020).
5. Bartlett, D. J. et al. High-Resolution Chest Computed Tomography Imaging of the Lungs. Investigative Radiology 54, 129–137 (2019).
6. Symons, R., Sandfort, V., Mallek, M., Ulzheimer, S. & Pourmorteza, A. Coronary artery calcium scoring with photon-counting CT: first in vivo human experience. The International Journal of Cardiovascular Imaging 35, 733–739 (2019).
7. Hsieh, S. S., Leng, S., Rajendran, K., Tao, S. & McCollough, C. H. Photon Counting CT: Clinical Applications and Future Developments. IEEE Transactions on Radiation and Plasma Medical Sciences 5, 441–452 (2021).
8. Yao, A. D., Cheng, D. L., Pan, I. & Kitamura, F. Deep Learning in Neuroradiology: A Systematic Review of Current Algorithms and Approaches for the New Wave of Imaging Technology. Radiology: Artificial Intelligence 2, e190026 (2020).
9. Chakrabarty, S. et al. MRI-based Identification and Classification of Major Intracranial Tumor Types by Using a 3D Convolutional Neural Network: A Retrospective Multi-institutional Analysis. Radiology: Artificial Intelligence 3, e200301 (2021).
10. Vaidyanathan, A. et al. Deep learning for the fully automated segmentation of the inner ear on MRI. Scientific Reports 11, 2885 (2021).
11. Ryu, K., Shin, N.-Y., Kim, D.-H. & Nam, Y. Synthesizing T1 weighted MPRAGE image from multi echo GRE images via deep neural network. Magnetic Resonance Imaging 64, 13–20 (2019).
12. Pinaya, W. H. L., Mechelli, A. & Sato, J. R. Using deep autoencoders to identify abnormal brain structural patterns in neuropsychiatric disorders: A large‐scale multi‐sample study. Human Brain Mapping 40, 944–954 (2019).
13. Bao, L. et al. Undersampled MR image reconstruction using an enhanced recursive residual network. Journal of Magnetic Resonance 305, 232–246 (2019).
14. Amoroso, N. et al. Deep Learning and Multiplex Networks for Accurate Modeling of Brain Age. Frontiers in Aging Neuroscience 11, (2019).
15. Díaz-Gómez, J. L., Mayo, P. H. & Koenig, S. J. Point-of-Care Ultrasonography. New England Journal of Medicine 385, 1593–1602 (2021).
16. Soni, N. J., & Lucas, B. P. (2015). Diagnostic point‐of‐care ultrasound for hospitalists. Journal of hospital medicine, 10(2), 120-124.
